Safety Where Every Process Is Regulated.
Cleanroom PPE compliance, chemical handling monitoring, GMP zone enforcement, and cross-contamination prevention — without slowing production or compromising quality.
Pharmaceutical and life sciences operations navigate the intersection of worker safety and product quality. Every process is regulated, every deviation documented, every contamination event costly. SAFVR's AURA engine monitors safety and compliance simultaneously — detecting PPE violations and handling errors in real time, automating GMP compliance workflows, training operators on zone-specific protocols, and predicting where compliance drift is emerging.
What Pharma Safety Leaders Face Today.
- 01
Safety Meets Quality
Worker safety violations in cleanrooms and production zones don't just risk injury — they risk contamination, batch rejection, and regulatory action against the product itself.
- 02
GMP Zone Discipline
Maintaining gowning compliance, zone boundary enforcement, and material transfer protocols across shifts without manual supervision at every entry point.
- 03
Chemical Handling Exposure
Hazardous material handling, solvent exposure, and reagent storage compliance require constant vigilance that human observation alone can't sustain.
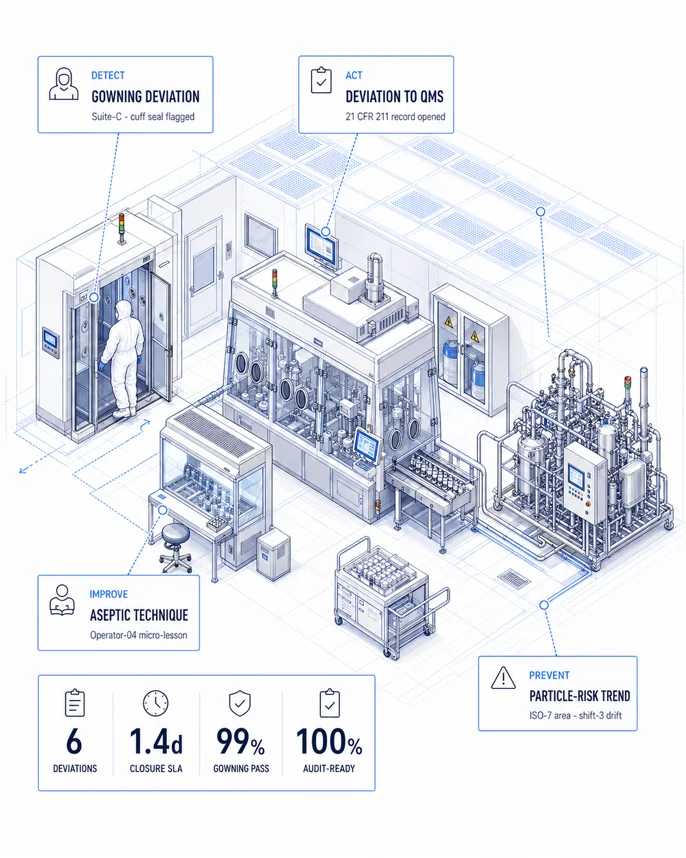
Detect. Act. Improve. Prevent.
- 01 / DETECT
Cleanroom & Production Zone Monitoring
Vision AI monitors cleanroom PPE gowning compliance, chemical handling procedures, GMP zone boundary enforcement, and cross-contamination risk indicators in real time.
Inside DETECT - 02 / ACT
GMP Compliance Automation
Automated deviation workflows triggered on detection — routing GMP zone violations to quality and safety teams simultaneously, with audit-ready documentation generated instantly.
Inside ACT - 03 / IMPROVE
Zone-Specific Operator Training
Micro-training modules calibrated to each GMP classification zone — covering the exact gowning, handling, and protocol requirements operators face in their assigned area.
Inside IMPROVE - 04 / PREVENT
Compliance Drift Detection
AURA tracks PPE compliance trends, zone violation frequency, and deviation close-out velocity across production lines — surfacing early indicators of quality and safety drift before audit findings.
Inside PREVENT
What Pharma Pilots Measure.
Outcomes vary by camera coverage, baseline maturity, and workforce mix. Pilot results are measured per site over the 30-day deployment window.
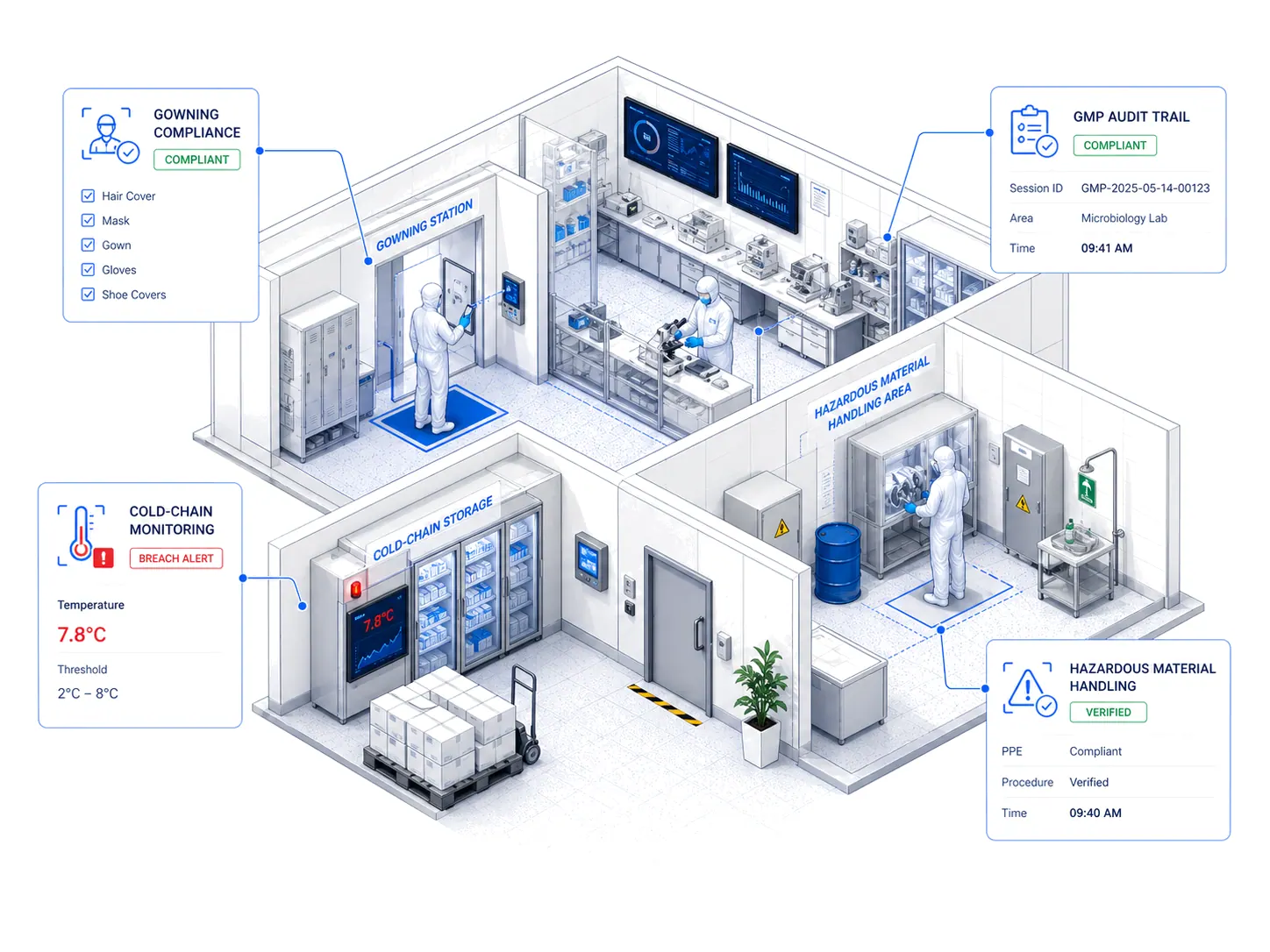
Every System. Every Zone. One Platform.
Cleanroom Gowning Compliance
AURA verifies the full gowning sequence at cleanroom entry points — detecting incomplete PPE, incorrect donning order, and zone-boundary breaches, with deviations routed to both quality and EHS teams.
GMP Zone Monitoring
Each GMP classification zone (A/B/C/D, ISO 5–8) is monitored for personnel flow, material transfer protocol, and environmental compliance — ensuring zone discipline without manual audits.
Cross-Contamination Prevention
Vision AI tracks material and personnel movement between classification zones, flagging breaches that could introduce contaminants — correlating events with batch records for root-cause traceability.
Chemical Handling Safety
AURA monitors chemical transfer, storage, and handling areas for PPE compliance, spill-risk postures, and unauthorised access — with every detection linked to the substance and zone for GHS documentation.
FDA Audit Evidence Generation
Continuous monitoring auto-generates the evidence trail FDA inspectors expect — timestamped deviations, corrective actions, training completion, and trend analysis — without manual log compilation.
Biocontainment Zone Discipline
Personnel entry, PPE compliance, and decontamination protocol adherence are monitored at BSL-classified zone boundaries — ensuring containment discipline is maintained across every shift.
Common questions about SAFVR for pharma & life sciences.
- How does AURA monitor gowning compliance in cleanrooms?
- Vision AI verifies gowning sequence, PPE completeness, and zone boundary discipline at every cleanroom entry — flagging deviations in real time to both quality and EHS teams with photographic evidence.
- Is AURA designed to support GMP and FDA inspections?
- AURA generates audit-ready documentation for every detection, deviation, and corrective action. Records are timestamped, evidenced, and retention-policy aware — designed to support GMP inspection readiness without additional manual log-keeping.
- Can AURA help prevent cross-contamination events?
- By monitoring material transfer protocols, zone-boundary breaches, and gowning discipline continuously, AURA surfaces the precursor patterns that lead to cross-contamination events — enabling intervention before batch impact.
- How does zone-specific training work for GMP classifications?
- Each GMP classification zone (A/B/C/D, ISO 5–8) gets its own micro-training module covering the exact gowning, handling, and protocol requirements operators face in that area — generated from your SOPs and recent deviation data.
- Does AURA slow down our production lines?
- No. AURA runs on existing cameras with no inline production interference. Detections, alerts, and documentation happen in parallel to production — observability without intrusion.
- Does SAFVR support cleanroom gowning compliance detection?
- Yes. AURA's vision AI is designed to support gowning sequence verification at cleanroom entry points — detecting incomplete PPE, incorrect donning order, and zone-boundary breaches in real time, with deviations routed to both quality and EHS teams.
Inside the Loop & Adjacent Industries.
Compliance That Doesn't Slow Production
Start a 30-day free pilot. AURA monitors your cleanrooms and production zones, proves GMP compliance improvements, and generates audit-ready evidence.